Treatments
Diclofenac Topical OTC Version Approved by the FDA

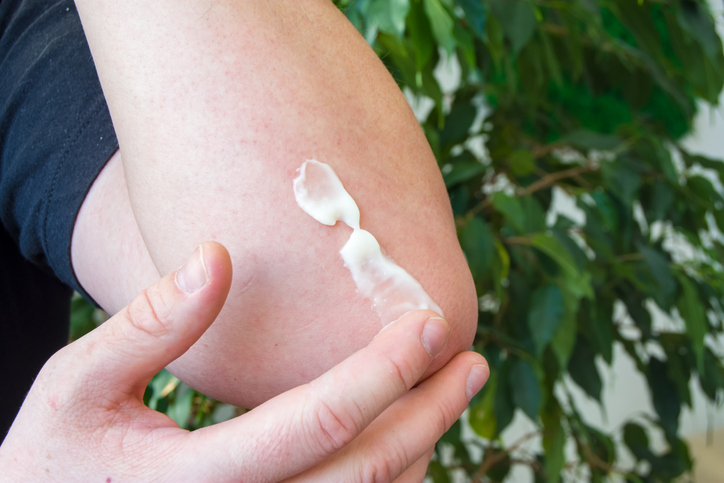
The U.S. Food and Drug Administration (FDA) approved diclofenac topical gel for over-the-counter (OTC) sale as of February 14, 2020. Previously, diclofenac (topical and oral) was FDA-approved only by prescription.
Diclofenac is a nonsteroidal anti-inflammatory drug (NSAID) that works by reducing body substances that cause pain and inflammation. Individuals who are considering using diclofenac topical gel should speak with their doctor or pharmacist to ensure that it does not interact with any other medication (oral or topical) or supplement (oral or topical) that they currently use. Use of diclofenac topical gel is not approved for those under the age of 18.
It may take up to seven days to feel any relief from diclofenac topical gel. If pain has not improved in seven days or if the need for use exceeds 21 days, individuals are urged to stop use and seek medical attention.
Instructions for use of diclofenac topical gel
- Diclofenac topical gel should only be used as directed and at the lowest effective dosage.
- It should only be used on the skin (external use only).
- If diclofenac gets in the mouth or eyes, they should immediately be rinsed with water.
- Diclofenac topical gel should never be applied to areas of the skin with infections, rashes, peeling, burns or open wounds.
- Diclofenac should be stored at room temperature and away from moisture and heat.
- If a dose is missed, the gel should be applied as soon as possible; however, if it’s almost time for the next dose, the missed dose should be skipped.